Current Projects
Electric field based tissue dissociation
Cell and Tissue Devices Team
Electrical tissue dissociation, pioneered and further developed in our lab, reframes tissue by applying tunable oscillating electric fields across tissue samples. This approach rapidly disrupts cell–cell and cell–matrix interactions to generate high-yield, viable single-cell suspensions without enzymes. Compared to conventional methods, electrical dissociation has demonstrated significantly improved cell recovery, preservation of native cellular states, and minimal transcriptomic perturbation, while enabling compatibility with automation through integrated device platforms. Further work is being done to increase the range of sample types and sizes that can be processed using these methods while maintaining the yield and lack of damage to cells.
Centrifuge
Mechanical Team
Designing and building a compact centrifuge for plug and play access on any liquid handler
Downstream Multi-Omic and Cellular Assays
Cell and Tissue Devices Team
To rigorously evaluate dissociation quality, we integrate downstream assays spanning flow cytometry, bulk DNA and RNA sequencing, and single-cell analysis workflows. Tissue dissociation is a critical determinant of data fidelity, influencing cell viability, population representation, and molecular integrity across assays. Optimized dissociation methods enable improved recovery of rare or fragile populations and better preservation of gene expression profiles, supporting high-quality flow cytometric phenotyping and reproducible genomic and transcriptomic measurements. Further work is being done to integrate spatial transcriptomics and immunofluorescence of intact tissues to compare to the same tissue post-dissociation.
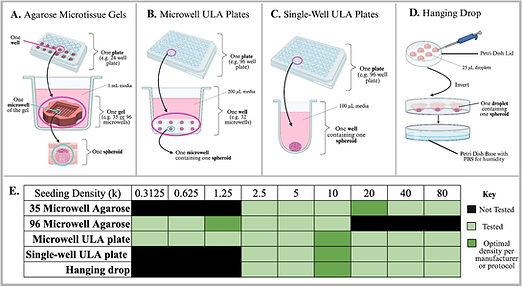
Organoid development
Cell and Tissue Devices Team
Building on advances in 3D culture systems, we investigate spheroid generation methods as a bridge toward more complex and physiologically relevant organoid models. Robust and reproducible formation of multicellular aggregates is essential for modeling tissue architecture, disease progression, and therapeutic response. This has progressed into development of endometrial organoids in collaboration with the Desai lab at Brown.